INDICATION
DYANAVEL® XR (amphetamine), Quillivant XR® (methylphenidate HCl), and QuilliChew ER® (methylphenidate HCl) are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in patients 6 years and older.
WARNING: ABUSE, MISUSE, AND ADDICTION
DYANAVEL XR, Quillivant XR and QuilliChew ER have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including DYANAVEL XR, Quillivant XR, QuilliChew ER, can result in overdose and death. Before prescribing DYANAVEL XR, Quillivant XR, QuilliChew ER, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout treatment, reassess each patient’s risk and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
- DYANAVEL XR, Quillivant XR, and QuilliChew ER are contraindicated:
-
- in patients known to be hypersensitive to amphetamine, methylphenidate, or other components of DYANAVEL XR, Quillivant XR, and QuilliChew ER. Hypersensitivity reactions, such as angioedema and anaphylactic reactions, have been reported.
- in patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of risk of hypertensive crisis.
- Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD doses. Serious cardiovascular effects with overdose may precipitate sudden cardiac death. Prior to treating patients with DYANAVEL XR, Quillivant XR, and QuilliChew ER, assess for the presence of cardiac disease. Avoid DYANAVEL XR, Quillivant XR, and QuilliChew ER use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease. Further evaluate patients who develop exertional chest pain, unexplained syncope, or arrhythmias during DYANAVEL XR, Quillivant XR, and QuilliChew ER treatment.
- CNS stimulants cause increase in blood pressure (mean increase approximately 2 to 4 mm Hg) and heart rate (mean increase about 3 to 6 bpm). Monitor all patients for tachycardia and hypertension.
- Use of CNS stimulants may cause exacerbation of pre-existing psychosis and may induce a manic or mixed episode in patients with bipolar disorder. In patients without prior history of psychotic illness or mania, CNS stimulants may cause new psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) at the recommended dosage. Prior to initiating DYANAVEL XR, Quillivant XR, and QuilliChew ER treatment, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing DYANAVEL XR, Quillivant XR, and QuilliChew ER.
- CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients with ADHD; monitor weight and height during treatment with DYANAVEL XR, Quillivant XR, and QuilliChew ER. Treatment may need to be interrupted in children not growing or gaining weight as expected.
- CNS stimulants including DYANAVEL XR, Quillivant XR, and QuilliChew ER are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; very rare sequelae include digital ulceration and/or soft tissue breakdown. Careful observation for digital changes is necessary during treatment with ADHD stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for DYANAVEL XR, Quillivant XR, and QuilliChew ER-treated patients who develop signs or symptoms of peripheral vasculopathy.
- CNS stimulants, including amphetamine and methylphenidate products have been associated with the onset or exacerbation of motor and verbal tics and worsening of Tourette’s syndrome. Before initiating DYANAVEL XR, Quillivant XR, and QuilliChew ER, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette’s syndrome. Discontinue treatment if clinically appropriate.
- Cases of priapism have been reported with methylphenidate use and during a period of withdrawal in both adult and pediatric male patients. Immediate medical attention should be sought in QuilliChew ER and Quillivant XR treated patients who develop abnormally sustained or frequent and painful erections.
- Serotonin syndrome risk is increased when DYANAVEL XR is co-administered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), MAOIs, and during overdosage situations. If it occurs, discontinue DYANAVEL XR and any concomitant serotonergic agents immediately, and initiate supportive treatment.
- QuilliChew ER contains phenylalanine, a component of aspartame, and can be harmful to patients with phenylketonuria (PKU). Before prescribing QuilliChew ER in patients with PKU, consider the combined daily amount of phenylalanine from all sources, including QuilliChew ER.
- Quillivant XR and QuilliChew ER treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
- Quillivant XR and QuilliChew ER should be prescribed to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Close monitoring of patients with a history of increased IOP or open angle glaucoma is necessary.
- Most common adverse reactions observed with amphetamine products: dry mouth, anorexia, weight loss, abdominal pain, nausea, insomnia, restlessness, emotional lability, dizziness, and tachycardia.
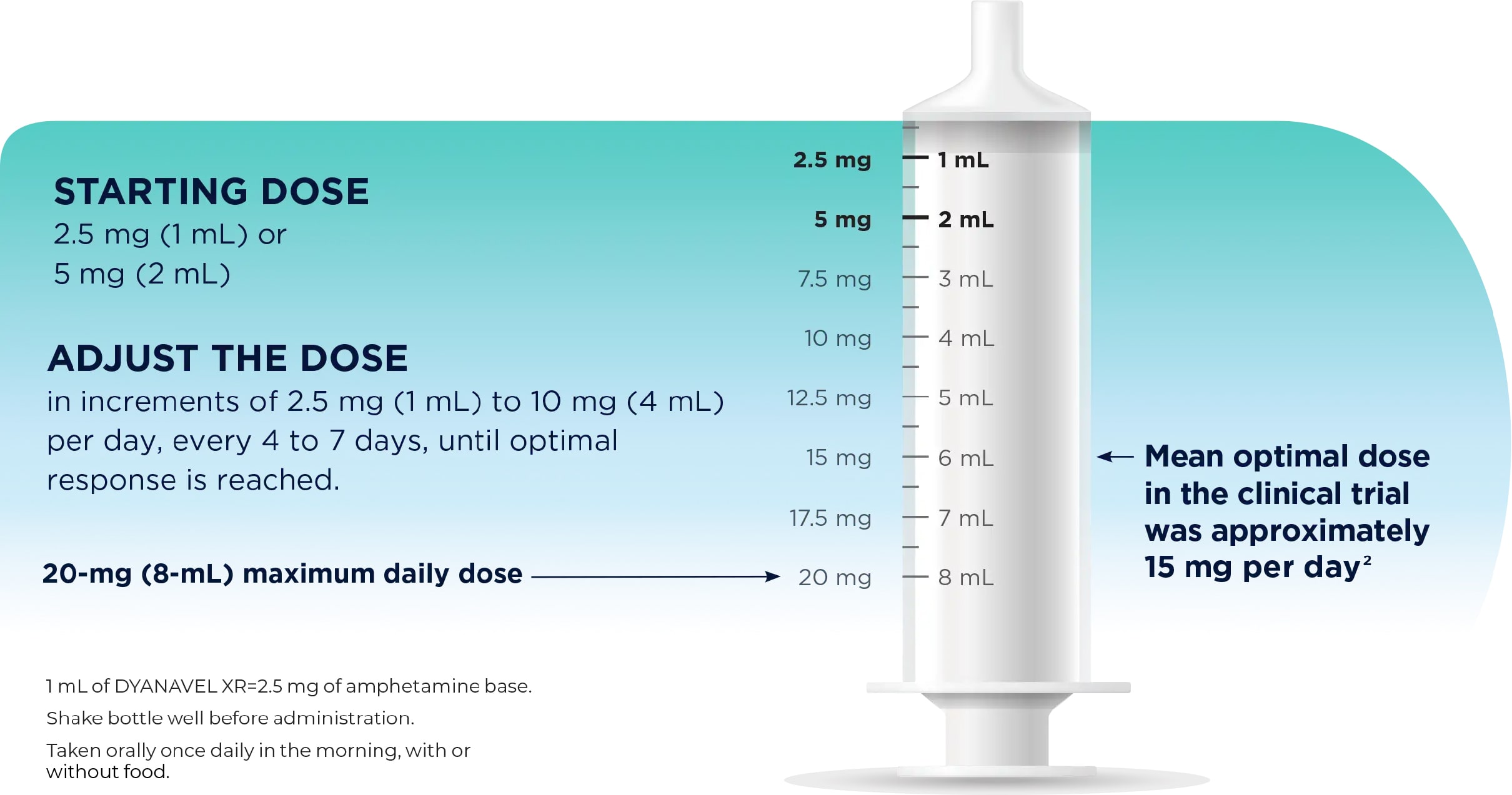
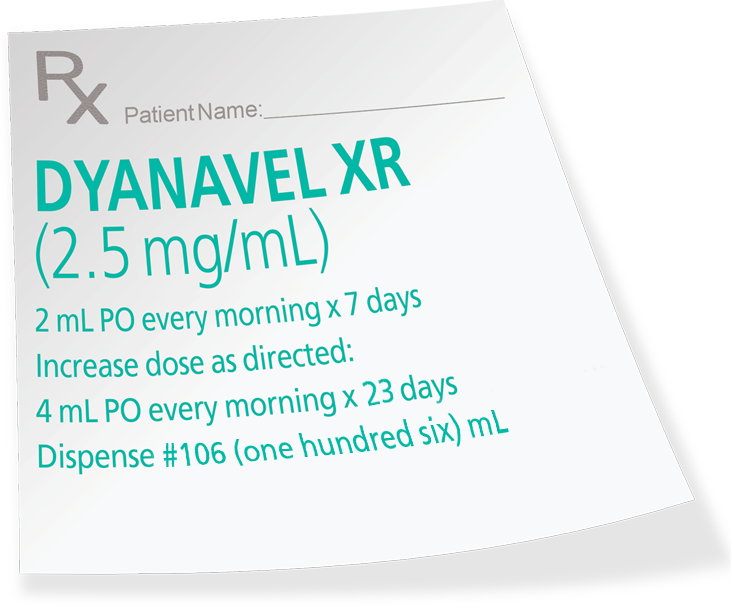
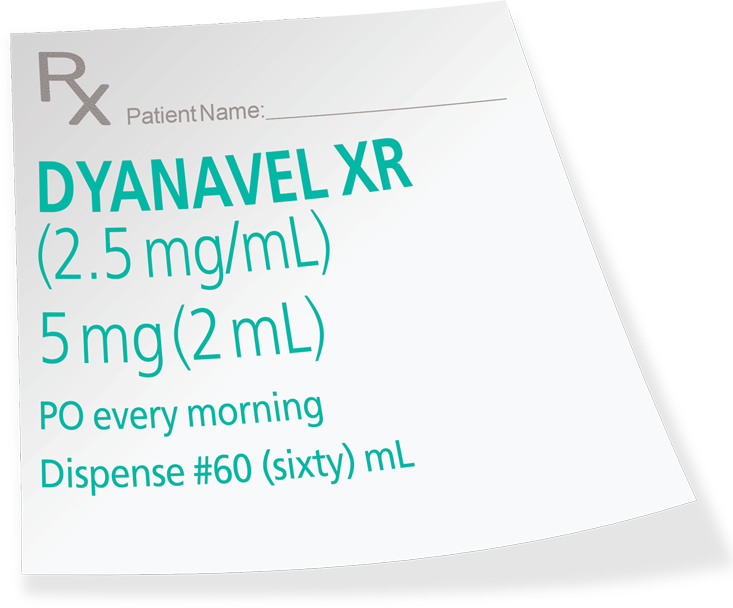
- Based on limited experience with DYANAVEL XR in controlled trials, the adverse reaction profile of DYANAVEL XR appears similar to other amphetamine extended-release products. The most common (≥2% in the DYANAVEL XR group and greater than placebo) adverse reactions reported in the Phase 3 controlled study conducted in 108 patients with ADHD (aged 6 to 12 years) were: epistaxis (DYANAVEL XR 4%, placebo 0%), allergic rhinitis (4%, 0%) and upper abdominal pain (4%, 2%).
- Based on accumulated data from other methylphenidate products, the most common (≥5% and twice the rate of placebo) adverse reactions are: appetite decreased, insomnia, nausea, vomiting, dyspepsia, abdominal pain, weight decreased, anxiety, dizziness, irritability, affect lability, tachycardia, blood pressure increased.
- There is limited experience with Quillivant XR and QuilliChew ER in controlled trials.
-
- Quillivant XR: The most common (≥2% in the Quillivant XR group and greater than placebo) adverse reactions reported in the Phase 3 controlled study conducted in 45 ADHD patients (ages 6 to 12 years) in Quillivant XR compared to placebo were affect lability (9% Quillivant XR, 2% placebo), excoriation (4%, 0%), initial insomnia (2%, 0%), tic (2%, 0%), decreased appetite (2%, 0%), vomiting (2%, 0%), motion sickness (2%, 0%), eye pain (2%, 0%), and rash (2%, 0%).
- QuilliChew ER: The most common (≥2% in the QuilliChew ER group and greater than placebo) adverse reactions reported in the Phase 3 controlled study conducted in 90 pediatric subjects (ages 6 to 12 years) in QuilliChew ER compared to placebo were decreased appetite (2.4% QuilliChew ER, 0% placebo), aggression (2.4%, 0%), emotional poverty (2.4%, 0%), nausea (2.4%, 0%), headache (2.4%, 0%), and weight decreased (2.4%, 0%).
- DYANAVEL XR use during pregnancy may cause fetal harm and because of the potential for serious adverse reactions in a breastfed infant, breastfeeding is not recommended during treatment with DYANAVEL XR.
- There are limited studies on the use of methylphenidate in pregnant women. However, premature delivery and low birth weight infants have been reported in amphetamine-dependent mothers. The developmental and health benefits of breastfeeding should be considered along with a mother’s clinical need for Quillivant XR and QuilliChew ER and any potential adverse effects on the breastfed infant from Quillivant XR and QuilliChew ER or from the underlying maternal condition. Monitor breastfeeding infants for adverse reactions, such as agitation, insomnia, anorexia, and reduced weight gain.
- To monitor pregnancy outcomes in women exposed to ADHD medications during pregnancy, healthcare providers are encouraged to register patients by calling the National Pregnancy Registry for Psychostimulants at 1-866-961-2388 or visiting online at https://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/othermedications/.
- To report SUSPECTED ADVERSE REACTIONS, contact Tris Pharma, Inc. at (732) 940-0358 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see Full Prescribing Information for DYANAVEL XR, Quillivant XR, and QuilliChew ER, including Boxed Warning regarding Abuse, Misuse, and Addiction.